
© 2026 Bao Nguyen Research Group. All Rights Reserved. Made by Tinh Nguyen

 > >
|
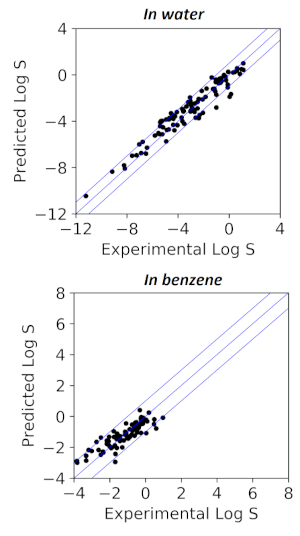
Research in Nguyen group focuses on understanding chemical processes and applying that knowledge to improve them for green chemistry and manufacturing. Some examples of recent projects are shown below: Solubility prediction for small molecules in organic solvents and water, with interpretable machine learning techniques. Using a combination of orthogonal physicochemical descriptors and reproducing established physical and chemical relationships, we achieved predictions approaching experimental errors and generality far outside the chemical space covered by the training data. This approach is named Causal Structure Property Relationship, and can provide much needed confidence in model performance with unseen data. See our paper on this at (doi:10.1038/s41467-020-19594-z).
|
|---|---|
|
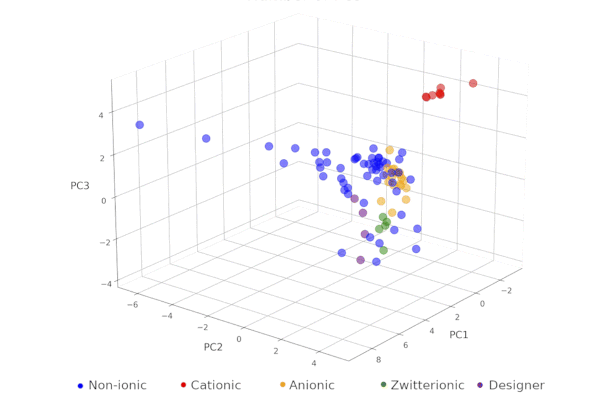
Surfactant map for surfactant selection in surfactant-enabled reactions in water. The surfactant map, which guides highly efficient surfactant screening to arrive at the optimal commercial surfactant for each individual organic reaction, is an extremely useful tool. It was built through Principal Component Analysis of 22 properties of 100 commercial surfactants, based on thorough investigations of the nature of these reaction systems. The reaction mixtures are complex emulsions where in partitioning of each reactant can affect the overall reaction performance. The map has been demonstrated to replace expensive designer surfactants with cheap, food-grade alternatives resulting in higher yields and/or easier purification. See our paper on this at (doi:10.1039/D3SC06311A). |
 > >
|
|
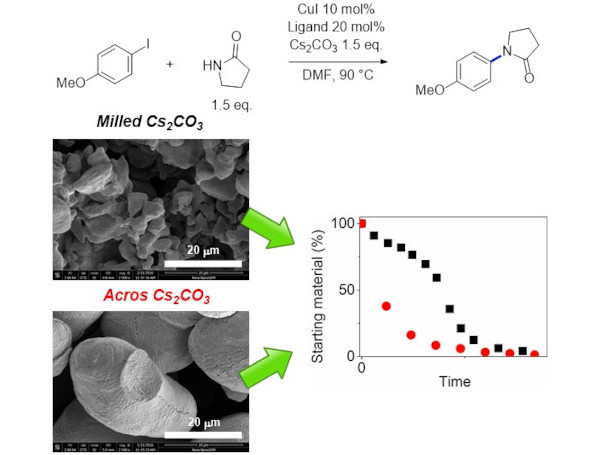
Identifying the origin of high catalyst loading and poor reproducibility of Ullmann-Goldber coupling reactions. These Cu-catalysed reactions are greener alternatives to Pd-catalysed C-N and C-O coupling reactions. However, most reactions requires 10 mol% of a copper(I) salt and 20 mol% of a N-N, N-O or O-O ligand. Industry also reported reactions failing upon scaling up, with lengthy induction periods. Our mechanistic investigation identifed ligand exchange as the main culprit behind both problems, and highlight potential solutions from a catalyst design perspective. See our paper on this at (doi:10.1039/C7SC02859H). |
 > >
|
Some of our recent research grants are listed below: